
Category: DYES AND CHEMICALS
Country: Netherlands
By Ada McVean
7th March 2024
Enzyme engineering could enable a greener and safer way to dye denim, with only a modest increase in cost. The method could alleviate the environmental and health impacts linked to the manufacture of jeans, of which around four billion pairs are sold each year.
Denim is given its typical blue colour by dyeing it with synthetic indigo in an industrial process that uses alkaline conditions and harsh reducing agents. ‘Currently we have a lot of pollution going out from the dyeing mills,’ explains Ditte Hededam Welner, a researcher at the Novo Nordisk Foundation Center for Biosustainability at the Technical University of Denmark. This pollution often negatively impacts local agriculture, she adds.
A dyeing process that uses indican – a compound closely related to indigo – may offer a greener alternative by eliminating the need for reducing agents. However, indican’s use has so far been hindered by the high cost of producing the compound.
Now, Hededam Welner and her colleagues have developed a chemoenzymatic process to produce indican that could make its use economically viable. The process converts indoxyl, a precursor to indigo used in existing dye manufacturing, into indican with the help of an engineered enzyme. By boosting the enzyme’s efficiency, while also improving the recyclability and minimising the costs of other required materials, the process reduces the cost of indican production to $5–13 per kilogram (£4–10 per kg), down from around $470 per kg. The final cost is comparable to the $5 per kg price of synthetic indigo.
Having established an economically feasible route to indican production, the researchers assessed the sustainability of two potential indican dyeing methods. The first relies on a natural β-glucosidase enzyme that can be sprayed onto indican wetted textiles to cleave indican’s β-O-glucoside bond and generate a radical indoxyl species. These radicals quickly dimerise to form indigo, providing the expected blue colour. Hededam Welner and her colleagues estimate the environmental impact of this enzymatic dyeing process as an order of magnitude lower than the current indigo-based method.
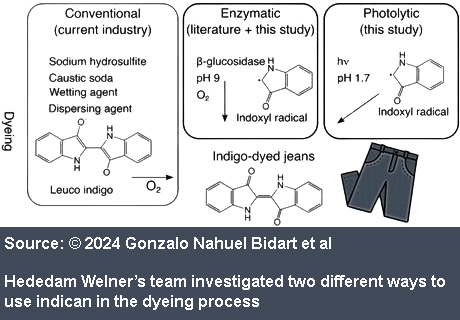 The second dyeing process uses light to trigger photolytic cleavage of indican, generating the indoxyl radicals. Effective dyeing of denim was possible using natural sunlight or a household light bulb, but it was most efficient with ultraviolet LEDs. While photolytic dyeing was also an improvement on current methods, the enzymatic dyeing process had a lower environmental impact overall , using less water and fewer toxic compounds.
The second dyeing process uses light to trigger photolytic cleavage of indican, generating the indoxyl radicals. Effective dyeing of denim was possible using natural sunlight or a household light bulb, but it was most efficient with ultraviolet LEDs. While photolytic dyeing was also an improvement on current methods, the enzymatic dyeing process had a lower environmental impact overall , using less water and fewer toxic compounds.
The team says that adopting its process would require relatively few changes to existing factories. However, the supply of indican is one major barrier to commercial use. ‘The indican needs to come from somewhere – it’s not currently produced in the world,’ says Hededam Welner. ‘You can get Sigma Aldrich to make it for you for lab use, but it’s not something you can buy in the tonne quantities that are needed for denim dyeing, so that whole production pipeline needs to be built.’
 Marco Fraaije, a biotechnologist at the University of Groningen in the Netherlands, points to consumers’ price expectations as another barrier. ‘The scale that that denim is produced is just almost unimaginable,’ he says. ‘The consumer is not willing to pay anything more than they are used to paying for it … as long as the prices are so low for jeans, it will be difficult to compete.’
Marco Fraaije, a biotechnologist at the University of Groningen in the Netherlands, points to consumers’ price expectations as another barrier. ‘The scale that that denim is produced is just almost unimaginable,’ he says. ‘The consumer is not willing to pay anything more than they are used to paying for it … as long as the prices are so low for jeans, it will be difficult to compete.’
‘This new process of going from indican is intriguing,’ comments Fraaije. ‘It goes back to the old-fashioned way of dyeing with indigo because that’s how they did it with the plants before we started with the chemical process.’
However, Fraaije is unsure if the new method can be implemented on a commercial scale, pointing to previous efforts to reduce the environmental impact of indigo dyeing. ‘It’s so difficult to predict which one will be the process that will run in the future,’ he says, although he notes that changes will have to be made eventually. ‘For sure in the future, maybe not in [the next] few years, but in 10 or 20 years, things have to change.’
References
G N Bidart et al, Nat. Commun., 2024, DOI: 10.1038/s41467-024-45749-3
Ada McVean
Ada has been Digital content assistant at Chemistry World since February 2023. A science writer since 2016, Ada has worked with the McGill Office for Science and Society, Skeptical Inquirer, SciMoms, Atlas Obscura and now Chemistry World.
Courtesy: Chemistryworld.com
Copyrights © 2026 GLOBAL TEXTILE SOURCE. All rights reserved.